|
|
| Yu-Jang Li |
 |
| Title:Professor, Chairperson |
|
Education: Ph.D., University of Rochester
Experiences :
1995/5-1996/6 Research associate, Rensselaer Polytechnic Institute.
1996/8-2003/7 Associate Professor, Chaoyang University of Technology.
2003/8-2011/7 Associate Professor, National Chiayi University.
2011/8- Professor, National Chiayi University.
2013/2-2018/1 Dean, Office of International Affairs, NCYU
2019/8- Chairperson of Department
|
| Research Area: Organic Chemistry, Asymmetric Synthesis |
| Tel: 886-5-271-7745
e-mail: yjli@mail.ncyu.edu.tw |
| Research Interests:
My research interest has been focused on the studies of ;
1. chiral [2,3]-Wittig rearrangement of g -allyloxy substituted vinylogous urethane enolates
2. [3,3]-Claisen rearrangement of g -allyloxy substituted vinylogous urethanes
.jpg)
3. Synthesis of biologically active natural products
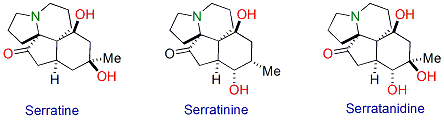
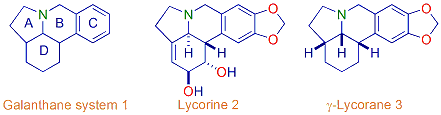
4. Total synthesis of indolizidine alkaloids
.gif)
 2014/09/16 2014/09/16
5. Synthesis of highly-substituted oxetanes
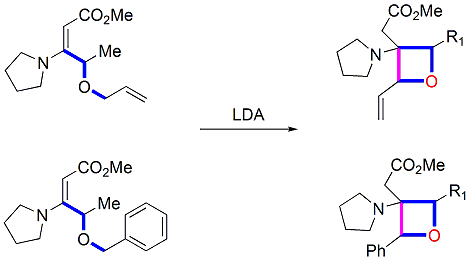
|
Publications:
- Synthesis of Isocondensed Hetereoaromatic Pyrroles. Sha, C. K.; Tsou, C. P.; Tsai, C. Y.; Liu, J. M.; Lee, R. S.; Li, Y. J.; Tsai, F. Y.; Way, S. J.; Young, J. J.; Chuan, K. S.; Yeh, R. H. J. Chinese, Chem , Soc . 1992 , 39 , 635., 1992年,(SCI)
- Total Synthesis of (-)Virginiamycin M2 Using Second-Generaton Vinylogous Urethane Chemistry, Schlessinger, R. H.; Li, Y. -J., J. Am. Chem. Soc . 1996 , 118 , 3301., 1996年04月,(SCI)
- Non-Racemic Syn-Selective Aldol Reaction with a Second-Generation Vinylogous Urethane Lithium Enolate., Schlessinger, R. H.; Li, Y. -J.; Von Langen, D. J., J. Org. Chem . 1996 , 61 , 3226., 1996年05月, (SCI)
- Birch Reduction and Reductive-Alkylation of 3,4-Dihydro-3-methyl -8-phenylisocoumarin, Schultz, A. G.; Li, Y. -J., Tetrahedron Lett ., 1996 , 37 , 6511., 1996年09月, (SCI)
- Birch Reduction of 3,4-Dihydro-8-hydroxy-3-methyl isocoumarin (Mellein) ...Expeditious Synthesis of (+-)-Ramulosin and a Sprce Budworm Toxin.Schultz, A. G.; Li, Y. -J., Tetrahedron Lett . 1997 , 38 , 2071., 1997年,(SCI)
- Novel Synthesis and g -Alkylation Reactions of 4-(1-Pyrrolidinyl)-2(5H)- Thiophenones., Yu-Jang Li, Zen-Ting Liu, and Sheng-Chuan Yang .. Tetrahedron lett .. 2001 , 42 , 8011. Supported by NSC-87-2113-M-324-004 (SCI)
- [2,3]-Wittig rearrangement of methyl b -pyrrolidinyl- g -alloxy-(E)-2-butenoate. Expeditious synthesis of 5-alkenyl-4-pyrrolidin-1-yl-5H-furan-2-ones, Yu-Jang Li, Pei-Ting Lee, Chuen-Mei Yang, Yuan-Kang Vhang, Ying-Chieh Weng and Yi-Hung Liu. Tetrahedron lett . 2004 , 45 , 1865-1868. Supported by NSC-92-2113-M-415-005 (SCI)
- An electrosprsy ionization tadem mass spectrometry based system with an online dual-loop cleanup device for simultaneous quantitation of urinary benzene exposure biomarkers trans,trans-muconic acid and S-phenylmercapturic acid. Rapid communication in mass spectrometry , Lin, L. -C.; Shih, J. -F.; Shih, T. -S.; Li, Y. -J.; Liao, B. -C. 2004 , 18 , 2743-2752. (SCI)
- A Glass Distilling collector applied for HCN Recovery from Submerged-Cultivated Broth and Fruiting Boby of Pleurotus eryngii for Identification and Quantification. Pei-Yu Chou, Chian-Huei Hong, Wenlung Chen, Yu-Jang Li, Yen-Shang Chen, and Robin Y. – Y. Chiou.* J. Agic. and Food Chem. 2006 , 54 , 1551-1556. (SCI)
- Recent Developments and Reviews of Reactions Related to The Pyrrolidine Substituted Vinylogous Urethanes, Chemistry, 2006, 64, 61-72.Yu-Jang Li*, Guo-Ming Ho.
- Asymmetric [2,3]-Wittig Rearrangement of The Dienolates of Chiral Secondary Alcohol Substituted ß-Pyrrolidinyl-g-Allyloxyl-a,b-Unsaturated Ester: Total Synthesis of (+)-Eldanolide.Yu-Jang Li,* Guo-Ming Ho, Pin-Zu Chen, Tetrahedron: Asymmetry 2009, 20, 1854-1563. (SCI) [Full Text Article-pdf]
-
Asymmetric [3,3]- and [1,3]-Sigmatropic Rearrangement of g-Allyloxy Vinylogous Urethanes. Yu-Jang Li,* Yuan-Kang Chang, Guo-Ming Ho, Hua-Ming Huang, Org. Lett. 2009, 11, 4224-4227. (SCI) [Full Text Article-pdf]
 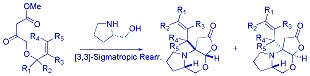
-
Anti-aldol reactions of chiral alcohol-substituted vinylogous urethanes and the synthesis of (-) Prelactone B. Yu-Jang Li,* Hsiu-Yin Hung, Yu-Wei Liu, Pei-Jhen Lin, Hung-Jyun Huang. Tetrahedron 2011, 67, 927-935.
-
Synthesis of Chiral Furopyrrolizidinones via a Key Tandem Cope Elimination/1,3-Dipolar Cycloaddition Reaction of Tricyclic Lactones, Yu-Jang Li,* Hsiang-Yu Chuang, Shang-Ming Yeh, and Wen-Sang Huang. Eur. J. Org. Chem. 2011(10), 1932-1939.
 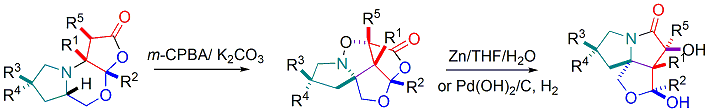
-
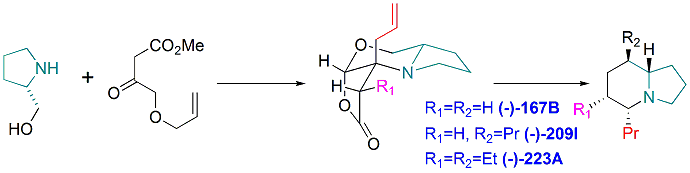
Total synthesis of Indolizidine Alkaloids (-)-167B, (-)-209I, and (-)-223A by Using a Common Tricyclic Lactone. yu-Jang Li,* Chung-Chien Hou, and Kuei-Chen Chang. Eur. J. Org. Chem. 2015(8), 1659-1663.
-
Stereoselective Synthesis of 2,3,4-highly Substituted Oxetanes by Intramolecular C-C Bond Forming Michael Addition, Guo-Ming Ho and Yu-Jang Li*. Chem. Commun, 2016, 52(81), 12108-12111. DOI: 10.1039/C6CC06857J. Supported by NSC 101-2113-M-415-002, MY2. Reviewed by V. Snieckus, B. P. Schuff on SYNFACTS, 2016, 12, 1242. " Stereoselective Preparation of Substituted Oxetanes"
 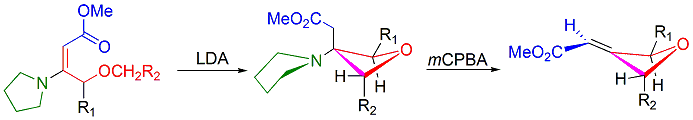
.
-
Discovery of New Stilbene Antioxidants of the Bio-Elicited Peanut Sprout Powder (BPSP) and Longevity Extension of Mice Fed with BPSP-Supplemented Diets. Robin Y.-Y. Chiou*, Po-Chang Chiu, Ju-Chun Chang, Yu-Jang Li, Chia-Wen Hsieh, Jin-Yi Wu, Shu-Mei Lin, Yun-Lian Lin, Brian B. C. Weng. Food and Nutrition Science, 2017, 8, 141-162.
 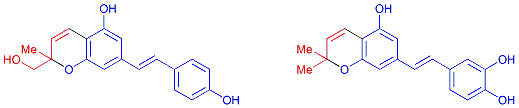
-
anti-Selective aldol reactions of chiral alcohol-substituted gama-benzyloxyl vinylogous urethanes and the synthesis of 3-benzyloxyl-4-hydroxylalkane-2-ones. Yu-Jang Li*, Chuan-Chung Chung, Pin-Zu Chen. Tetrahedron: Asymmetry 2017, 28, 1573-1581. https://doi.org/j.tetasy.2017.10.003.
 .gif)
-
Tandem [1,2]-Wittig Rearrangement/Lactonization of gama-benzyloxy vinylogous urethanes: Application to the Synthetic studies of Maculalactone A, Planchol C and gama-Lycorane. Guo-Ming Ho, Yu-Jang Li*. Asian J. Org. Chem. 2018, 7, 145-149.
 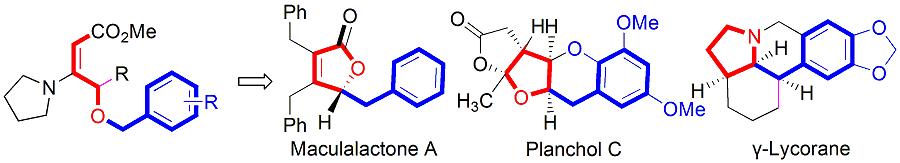
-
Peroxidase characterization isolated from germinated peanut embryos (GPE) and application of the freezed-dried GPE powder as emzyme source for biomimetic production of delta-viniferin. Po-Chang Chiu, Yu-Jang Li, Robin Y.-Y. Chiou*. J. Procbio. 2018, 66, 97-105. DOI: 10.1016/j.procbio.2017.11.019.
 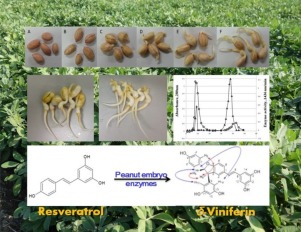
-
C-H Functionalization of Amino Alcohols by Osmium Tetroxide/NMO or TPAP/NMO: Protection Group-Free Synthesis of Indolizidines (-)-223AB and 3-epi-(-)-223AB. Wei-Lun Chen, Lee-Ya Wang, Yu-Jang Li*. Eur. J. Org. Chem. 2020, 103-107. http://dx.doi.org/10.1002/ejoc.201901494.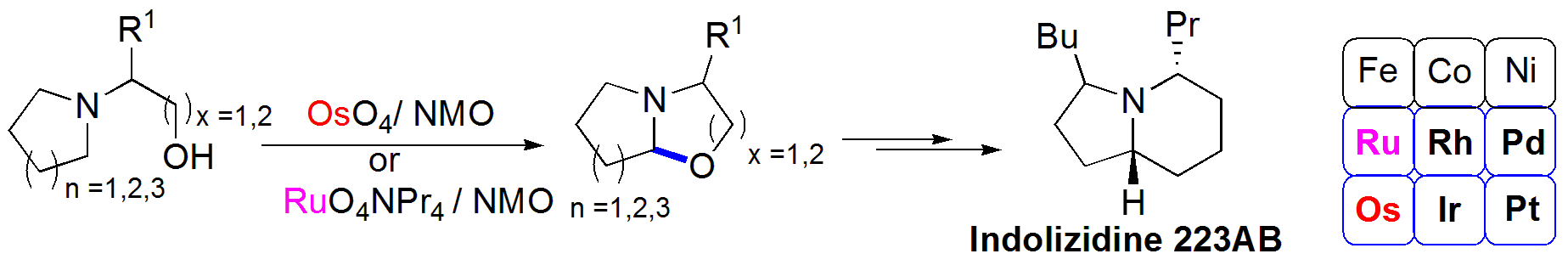
-
Inhibition of testosterone-mediated benign prostatic enlargement of orchiectomized Sprague-Dawley rats by diets supplemented with bio-elicited peanut sprout powder (BPSP) and three new BPSP-extracted natural compounds identified. Cheng, P.-H., P.-C. Chiu, J.-C. Chang, S.-M. Lin, Y.-J. Li, D.-Y. Lo, L.-R. Lai, S.-C. Wu and R. Y.-Y. Chiou*. J. Functional Foods, 2021.79 (2021) 104383.

| |

 |
|